Charlize
|
Manufacturer
Distributor Contents Indications Dosage Administration Contraindications Special Precautions Adverse Drug Reactions Side Effects Drug Interaction Storage Description Mechanism of Action MIMS Class ATC Classification Regulatory Classification |
Pond's chemical
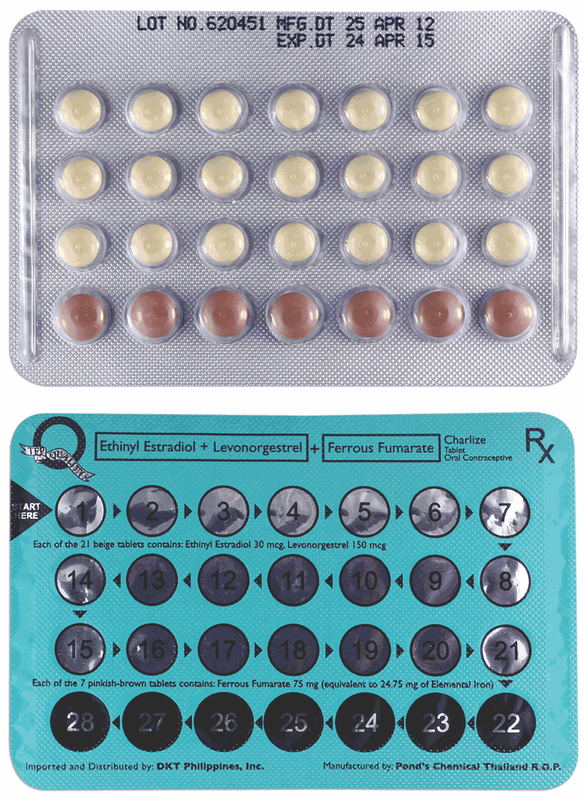
DKT Ethinyl estradiol 30 mcg, levonorgestrel 150 mcg, Fe fumarate 75 mg Contraception and also for menstrual disorders eg, dysmenorrhea, premenstrual syndrome and menorrhagia. 1 tab daily starting on the 1st day of menstrual cycle for 28 days. Should be taken on an empty stomach: Best taken on an empty stomach. May be taken w/ meals to reduce GI discomfort. Women with liver disorders, clotting disorders, breast and cervical cancer, sickle-cell anemia, hormone-active tumors, hyperlipidemia, severe cardiovascular diseases, previous or existing thromboembolic diseases and idiopathic jaundice. Use in pregnancy: Prior to starting Charlize, pregnancy must be ruled out. However, should a pregnancy occur while taking the tablet, the administration has to be withdrawn at once. Use in lactation: Women with newborn babies should not use Charlize when breastfeeding. Before taking hormonal contraceptives, the woman should undergo an appropriate medical examination and her medical history should be carefully evaluated. Regular examination is recommended during use. The contraceptive effectiveness of combined preparations may be reduced during episodes of vomiting or diarrhea. Using a condom is recommended for 7 days after recovery to avoid pregnancy during this period. Consult a healthcare provider if suffering from the following conditions: Diabetes mellitus, high blood pressure, varicose veins, multiple sclerosis, epilepsy, migraine, ophthalmological problems and tetany. Headaches, gastric upsets, nausea, vomiting, impaired appetite, breast tenderness, slight changes in body wt & libido, depressive moods or interference w/ liver functions. The oral contraceptive is the most researched product in the history of modern medicine and its safety has long been established. Some women may experience side effects eg, headaches, gastric upsets, nausea, vomiting, impaired appetite, breast tenderness, slight changes in body weight and libido, depressive moods, or interference with liver functions. Hepatic microsomal enzyme inducers including barbiturates eg phenobarb, primidone, carbamazepine, oxcarbazepine, felbamate & topiramate, griseofulvin, phenylbutazone, phenytoin, troglitazone, St. John's wort & rifampicin may decrease contraceptive efficacy &/or increase breakthrough bleeding. Anti-infective agents eg ampicillin, chloramphenicol, neomycin, nitrofurantoin, penicillin V, sulfonamides & tetracyclines may decrease contraceptive efficacy. Troleandomycin may increase risk of cholestatic jaundice. CYP3A4 inhibitors eg clarithromycin, erythromycin, grapefruit juice, itraconazole, ketoconazole, ritonavir may increase plasma conc of ethinyl estradiol. Analgesics, INH, antimigraine drugs & tranquilizers may decrease contraceptive efficacy. Store at temperatures not exceeding 30°C. Shelf-Life: 36 months. Each of the 21 beige tablets contains ethinyl estradiol 30 mcg and levonogestrel 150 mcg. Each of the 7 pinkish-brown tablets contains ferrous fumarate 75 mg (equivalent to 24.75 mg of elemental iron). Pharmacotherapeutic Group: Oral contraceptive. Charlize is a low-dose monophasic contraceptive pill. Each tablet contains a minimum amount of contraceptive substances, thus, the risk of side effects is greatly reduced. Ferrous fumarate is an iron supplement that helps improve the hemoglobin content of blood during menstruation. Charlize is a convenient and simple-to-use contraceptive method that provides continuous protection. It has no effect on a woman's fertility. Also, it does not increase the risk for fetal abnormalities when a woman wants to have a baby later. When the patient decides to conceive, the patient simply has to stop taking the pill and the body will be ready for conception within weeks. A patient can take Charlize for as long as the patient wants. Pharmacokinetics: Ethinyl estradiol is absorbed well and rapidly by the GIT. The presence of an ethinyl group at the 17-position greatly reduces hepatic first-pass metabolism compared with estradiol, enabling the compound to be much more active if taken orally. There are some initial conjugation at the gut wall and the systemic bioavailability is only 40%. Ethinyl estradiol is highly protein bound, unlike naturally occurring estrogens, which are mainly bound to sex hormone-binding globulin; it is principally bound to albumin. It is metabolized in the liver and excreted in urine and feces. Metabolites undergo enterohepatic recycling. Levonorgestrel is rapidly and almost completely absorbed after being taken orally and undergoes little first-pass hepatic metabolism. It is highly bound to plasma proteins; 42-68% to sex hormone-binding globulin and 30-56% to albumin. The proportion bound to sex hormone-binding globulin is higher when it is given with an estrogen. Levonorgestrel is metabolized in the liver to sulfate and glucuronide conjugates, which are excreted in the urine and to a lesser extent, in the feces. Oestrogens, Progesterones & Related Synthetic Drugs / Oral Contraceptives G03CA01 - ethinylestradiol ; Belongs to the class of natural and semisynthetic estrogens used in estrogenic hormone preparations. Rx |